Another challenging year is behind us. Political, socio-economic and climatic changes stimulate the development of science and technology on a daily basis and determine new trends. The world of chemistry has also changed during this time.

TOP 10 2023
We present 10 interesting discoveries and events that changed the world of chemistry in 2023 .

First hexazine compound [03.2023]
An article published in early March in Nature Chemistry reported on the synthesis of a new nitrogen compound. Researchers from the UK, China, Sweden and Germany have obtained an aromatic ring structure with the formula K9N56. The compound was formed by reacting nitrogen with KN3 in a laser-heated diamond anvil cell under high pressure (46 and 61 GPa) and temperature (over 2000 K) conditions[1].
The observed structure had, e.g., hexazine rings in the form of [N6]4-anions. They follow Hückel’s aromaticity rule. In addition, the macromolecular compound also contained flat N5 rings and N2 dimers.
So far, a six-member nitrogen ring called hexazine has only been theoretically proposed as a chain. The aromaticity of this element was limited to the pentazolate form [N5]–. The aforementioned team of scientists is the first to boast the synthesis of the macromolecular complex compound K9N56, which has a [N6]4-anion in its structure..
Miracle drug developed for deadly poisonous fungus species [05.2023]
Amanita phalloides, commonly known as death cap, is one of the most toxic mushroom species for humans. It is responsible for about 90% of all fatal poisonings from mushroom consumption. The most potent toxin of the fungus, responsible for permanent damage to the liver and other organs in humans, is α-amanitin. Even aggressive treatments for ingestion of this toadstool are sometimes ineffective in most cases.
Nevertheless, the study, published in May in the Nature, brings hope in the future fight against the death cap poisoning. The team of researchers, using the CRISPR method, created a pool of cells with different mutations, observing what factors might affect their resistance to α-amanitin. The results of the experiment indicated that cells in which the STT3B protein was absent were not destroyed by the toxin. Among the known approved medical substances, the dye – indocyanine green (ICG) – has been proposed as a specific STT3B inhibitor. The compound is typically used in medical imaging and could be a potential specific antidote for α-amanitin poisoning. Experiments on mice showed that administration of indocyanine green four hours after ingesting the fungus significantly increased survival and protected them from the toxin, stopping liver damage[2].
Methyl cation observed in space [06.2023]
The chemical properties of the element carbon have been the subject of much research over the years. Observations in this area bring us closer to understanding the mysteries of the evolution of life on Earth, as well as the possibility of its development elsewhere in the Universe.
In June, an international team of scientists released an image taken with NASA’s James Webb Space Telescope. The image depicted a new, previously unobserved carbon compound – the methyl cation CH3+. The particle is located in a star system about 1,350 light years away from us – the Orion Nebula.
UV radiation in space has a degrading effect on most organic structures. However, scientists speculate that this energy has an important role in methyl cation formation. The ion, once formed, undergoes further chemical reactions to build more complex carbon molecules – traces of life[3].

Scientists have determined the structure of boron monoxide [07.2023]
The chemical compound with the molecular formula BO was first proposed in the 1940s. Still, it was impossible to determine its structure due to limited access to technology at that time. Current scientific interest in flat boron structures has once again drawn attention to the oxide described almost a century ago.
Advanced NMR (Nuclear Magnetic Resonance) spectroscopy analysis methods helped Frédéric A. Perras’ team determine the most likely orientation of the boron oxide particles. The Ames scientists observed that the precursor molecules in the reaction arranged themselves in a parallel fashion to each other, forming the so-called 2D “nanosheets” composed of B4O2 rings with oxygen bridges. As a result of powder diffraction studies, it was further postulated that these units form layers with irregular stacking patterns. According to F.A. Perras – the layout resembles a stack of paper thrown on a desk – slightly disorganized, but remaining in its form. The measurements made coincide with preliminary assumptions of the BO structure estimated in 1961[4].
Cyclocenes – new cyclic structures [08.2023]
The branch of organometallic chemistry focuses on organic compounds that have at least one bond between a metal and carbon atom. Among the various structures, there are complexes that layered in a specific way. The first such compound discovered was ferrocene. Using its example, it is easy to understand the structure of organometallic complexes, the central atom – a metal – is surrounded by a group of ligands connected to it.
In 2023, Peter Roesky’s team at Karlsruher Institut für Technologie developed and described a new type of complexes called “cyclocenes.” These compounds can contain up to 18 layer units. Cyclocenes have cyclooctatetraene (COT) ligands, with two silane groups each attached thereto. COT ligands surround metal (e.g. strontium) centers by arranging themselves in a characteristic ring around the atom. Scientists are pinning their hopes on the usefulness of the new compounds in the future of organometallic chemistry[5].
The most waterproof surface ever [10.2023]
Hydrophobicity means the ability of a material to repel water molecules. Waterproof coatings are widely used in numerous areas of life. In order to meet increasing material requirements, research in the field of nanotechnology over the past three decades has contributed to the development of numerous advanced hydrophobic structures.
Using a specialized reactor, a team of researchers from Aalto University in Finland, has proposed a brand new form of “liquid-like omniphobic surfaces.” Their work represents the first instance of experiments in this area, at the extremely detailed nanoparticle level. The aforementioned surfaces have covalently bonded, substrate-bound molecular layers with the simultaneous ability to be highly mobile. Their properties can be likened to a layer of lubricant between the water droplets and the surface itself.
Among other existing structures – in terms of characteristics, this one representsthe most slippery and fluid surface in the world. It is estimated that omniphobic nanoparticles could find applications, e.g., in heat transfer processes in pipes and systems, de-icing of surfaces, and prevention of evaporation. Promising potential for the developed surfaces can also be seen in the field of microfluidics[6].

Leonardo da Vinci’s chemical experiments [10.2023]
Leonardo da Vinci became famous as one of the most recognizable figures living during the Renaissance. The artist had many talents, which he transformed into timeless works of art or inventions. Recent analyses of microsamples taken of the paints used in his works, the Mona Lisa and The Last Supper, indicate that da Vinci not only painted beautifully, but also experimented effectively in obtaining the best properties of his components.
An international team of scientists pointed to the presence of toxic lead compounds in the base layer of both paintings. The painter probably tried to apply thick bands of white lead pigment by additionally saturating the oil used on the canvas with lead oxide (PbO). Using X-ray diffraction and infrared spectroscopy techniques, the researchers determined that the paintings contain not only lead white, but also a much rarer compound: plumbonacrite (Pb5[CO3]O[OH]2) – which is stable only in alkaline environments. This discovery may indicate that Leonardo pioneered this painting technique[7].

Nobel Prize in Chemistry 2023 [10.2023]
Moungi G. Bawendi, Louis E. Brus and Alexei I. Ekimov won the 2023 Nobel Prize in Chemistry. It was the 115th Nobel Prize in Chemistry, awarded by the Royal Swedish Academy of Sciences, which honored scientists “for the discovery and synthesis of quantum dots.”
Quantum dots are a part of nanoparticle group whose size is small enough to determine their characteristic features. The function of quantum dots is based largely on the emission and absorption of radiation. The unique electronic, as well as optical properties of these nanoparticles, allow for a wide range of applications, including photovoltaic devices, screens of certain TV sets or LED lamps.
After mentioning a few pieces of information about quantum dots, it is now time to tell the story of the Nobel Prize winners. Scientist Alexei Ekimov of Nanocrystals Technology Inc. first synthesized quantum dots in a glass matrix in 1981. Two years later, Louis Brus of Columbia University, obtained the same structure, yet in a colloidal suspension. On the other hand, Moungi G. Bawendi of the Massachusetts Institute of Technology, along with a team of researchers, have developed one of the most popular and widely used methods for synthesizing quantum dots to produce near-perfect molecules[8].

2023 “Polish Nobel Prize” [11.2023]
The Foundation for Polish Science awards were presented for the 32nd timein 2023. The award is often referred to as the “Polish Nobel Prize”, as it is considered the most important scientific award in Poland. It is awarded to “outstanding scholars for special scientific achievements and discoveries that push the boundaries of cognition, open up new research perspectives, make outstanding contributions to the civilizational and cultural progress of our country, and ensure its prominent place in addressing the most ambitious challenges of the modern world.”
This year’s Award in the field of chemical and material sciences went to Prof. Marcin Stępień of the Department of Chemistry at the University of Wroclaw. The award was given “for designing and obtaining new aromatic compounds with unique structures and properties.”
The concept of “aromaticity” has been known in chemistry since the second half of the 19th century, and has been developed all the time thanks to scientists, such as Professor Stępień. The fruit of his scientific work was the design and synthesis of new aromatic and anti-aromatic molecules, which were distinguished by their unique structure and unusual, often three-dimensional shapes. These are important achievements from a cognitive point of view, but also open up new applications for these compounds as functional organic materials. The molecules synthesized by the award-winning professor may inspire the search for new organic materials (especially functional dyes), which may find applications in photovoltaic and LED devices, or in phototherapy and medical diagnostics, among others[9].
Liquid metals in catalyst production [11.2023]
November 2023 brought good news from a team of scientists in Australia, who discovered that liquid metals can be used to produce catalysts. So far, they have mostly been produced from solid materials, including metals or organometallic compounds. Nevertheless, their use required high temperatures, resulting in increased energy consumption and greenhouse gas emissions.
A group of researchers led by Professor Kourosh Kalantar-Zadeh studied the feasibility of using liquid metals (such as gallium), which can be catalysts at lower temperatures. This would reduce energy consumption and thus reduce greenhouse gas emissions in the chemical sector. This is very important because the industry is responsible for a significant portion of global emissions. In addition, the greater flexibility of liquid metals, compared to solid metals, provides the opportunity to improve the performance of catalysts.
The scientists in Australia plan to continue their research. If their discovery would be feasible, it could lead to the introduction of new, greener processes in chemical production[10].
[1] https://www.nature.com/articles/s41557-023-01148-7
[2] https://www.nature.com/articles/s41467-023-37714-3
[3] https://www.nasa.gov/feature/goddard/2023/webb-makes-first-detection-of-crucial-carbon-molecule
[4] https://pubs.acs.org/doi/abs/10.1021/jacs.3c02070
[5] https://www.nature.com/articles/s41586-023-06192-4
[6] https://www.nature.com/articles/s41557-023-01346-3
[7] https://pubs.acs.org/doi/10.1021/jacs.3c07000
[8] https://www.nobelprize.org/prizes/chemistry/2023/press-release/
[9] https://www.fnp.org.pl/rozmowy-z-laureatami-nagrod-fnp-2023/
[10] https://www.nature.com/articles/s41565-023-01540-x
TOP 10 2022
Here we present 10 interesting discoveries and events that changed the world of chemistry in 2022.
Scientists learned the weight of the neutrino (February 2022)
The existence of the neutrino was theoretically predicted by Wolfgang Pauli in 1930. This particle was supposed to balance the energy of the so-called radioactive beta decay. Despite various theories, it was impossible to confirm the existence of this particle for a long time, although with time it was called the neutrino.It is characterized by the fact that it has no electric charge and interacts very weakly with other particles.Some scientists assumed that the neutrino had no mass, just like the photon, while others thought it was simply very small.
Weighing a neutrino would allow us to understand the universe even better, which is why it became the subject of research in the international KATRIN project, led by the Karlsruhe Institute of Technology.In their experiments, scientists used the phenomenon of beta decay, which occurs in the atoms of a hydrogen isotope (called tritium).The KATRIN is a specialized research apparatus, its length is 70 m, and it includes a huge spectrometer, used to measure the properties of electrons generated during radioactive decay. The research has been ongoing since 2019 and brings better and better results. One of the results of this work is the determination of the mass of the neutrino, which is no more than 0.8 eV. For comparison, the mass of an electron is 0.511 MeV (million electron-volts) and the mass of a neutron is 0.938 GeV (billion electron-volts). This is the first time that the measurements succeeded below the electron-volt limit, which is why this is such an important achievement.[1]
Research under the KATRIN project is still ongoing and is expected to run until at least 2024. Scientists hope to obtain an even more precise result of measuring the mass of the neutrino.[2]

Carbon sink ( April 2022)
Mass-produced plastics can be found virtually everywhere today. One of the well-known products are plastic bottles, which can consist of several different materials, and their recycling is not the easiest. Although the recovery of plastic is associated with costs and it is a complicated process, which makes it still not very popular, scientists are looking for applications for the material recovered that could make this process even more beneficial.
A group of chemists from Rice University have announced the results of their research, which prove that used plastic can be helpful in the fight against high carbon dioxide emissions. As reported in the “ASC Nano”[3], journal, scientists have explored the process of pyrolysis, which is the breakdown of chemicals. It consists in heating particular substances to very high temperatures, maintaining anaerobic conditions. Currently, pyrolysis is used in the petrochemical industry, among others.
The chemists from the Rice University pyrolysed the plastic in the presence of potassium acetate, resulting in very specific molecules that have microscopic pores and are great at capturing and binding CO2 molecules. This material could be used as an ideal carbon dioxide absorber, e.g. in the form of filters for the chimneys of power plants burning fossil fuels. Such an absorber would be characterized by properties that allow for multiple use, and additionally, capturing a ton of CO2 with it would be several times cheaper than current methods of sequestration (capturing) of carbon dioxide.[4]

Quantum Nanomagnet (April 2022)
A quantum nanomagnet with unique properties is a discovery of scientists from the Jagiellonian University. A team of researchers led by Dr Dawid Pinkowicz, described in the “Nature Communications”[5], journal a new type of organometallic quantum nanomagnet, in which the central magnetic ion is surrounded only by other metal ions. The molecule consists of a central erbium ion, which combines with three heavy rhenium ions. This combination made it possible to get closer to the properties of the already known large macroscopic magnets.
Scientists emphasize that although molecular magnets will not be used in the near future, they can revolutionize the future and change such fields as electronics and computer science. Currently known molecular magnets require strong cooling; therefore, to find practical application for nanomagnets it is necessary to create them in such a way that they can operate at room temperature. Scientists await further research in this area.[6]

Sodium batteries (June 2022)
Lithium-ion batteries are used in most everyday devices. The production of such devices require rare metals such as cobalt and lithium, which is also not a common element and has a significant impact on the production price. According to the scientists, lithium could be replaced by sodium, which would significantly reduce production costs. In addition, sodium batteries would charge much faster and depleting the battery “to zero” would not have a harmful effect. However, the work carried out so far has ended in a fiasco, because sodium very quickly forms thin, metallic structures on the electrode – the so-called dendrites, which resulted in the short life of such batteries.
Researchers from the University of Texas in Austin solved this problem by using a computer model to create a new material that prevents the formation of dendrites and, as a result, it prevents damage to the electrode. It was made by depositing a thin layer of sodium on antimony telluride and folding it many times, thus creating alternating layers. Thanks to this, sodium is very evenly distributed and dendrites form much slower and less frequently on it. This allows to create a sodium battery that will match the lithium one in terms of the number of charge and discharge cycles, and will also have a comparable energy density. Sodium batteries may become the future for the industry.[7]

Nobel Prize in Chemistry 2022 [October 2022]
This year, the Royal Swedish Academy of Sciences awarded the Nobel Prize in Chemistry to three people. Carolyn R. Bertozzi, Morten Meldal and K. Barry Sharpless are the winners of the Prize, who were recognized “for the development of click chemistry technology and bioorthogonal chemistry”.
The “click chemistry” is a process that can be compared to a building made of LEGO bricks. Namely, specific fragments of molecules can be combined with each other to produce compounds of high complexity and diversity. The combination of simple elements, which we can call “chemical building blocks”, allows you to create an almost infinite variety of molecules. Bioorthogonal chemistry, on the other hand, allows to monitor the chemical processes that take place in living cells without damaging them. This offers a unique opportunity to study diseases inside cells as well as in complex organisms.
Both the “click chemistry” technology and bioorthogonal chemistry are important discoveries, mainly for medicine and pharmacy, which can significantly affect the development of both these fields.[8]
You can read more about the winners and awarded discoveries in the article “The Nobel Prize in Chemistry”, which is also available on the PCC Group blog.

“Polish Nobel Prize” 2022 (November 2022)
For the 31st time the Foundation for Polish Sciences awarded its prizes, which are considered the most important scientific prize in Poland and often also referred to as the “Polish Nobel Prize”. These prizes are granted for special discoveries and scientific achievements that push the boundaries of cognition, as well as opening new cognitive perspectives and making an outstanding contribution to the civilization and cultural progress of our country, and ensuring Poland’s significant place in taking up the most ambitious challenges of the modern world. The winner of this year’s Award in the field of chemical and material sciences was profBartosz Grzybowski.
Professor Bartosz Grzybowski from the Institute of Organic Chemistry of the Polish Academy of Sciences in Warsaw and Ulsan National Institute of Science and Technology in the Republic of Korea, was awarded “for the development and empirical verification of an algorithmic methodology for planning chemical synthesis”. His discovery consisted in carrying out a computer-planned organic synthesis and using artificial intelligence to predict the course of chemical reactions, and, at the same time, discover new compounds that could be used as medicines.
Professor Grzybowski is one of the first scientists in the world dealing with organic chemistry who decided that it was time to use the possibilities of computational methods and who developed tools that could predict not only real-life applicable, but also even better ways to synthesize difficult organic molecules.[9]
It is also worth mentioning the winner of the Award in the field of life and earth sciences – Professor Marcin Nowotny, who was awarded “for explaining the molecular mechanisms of DNA damage recognition and repair”. The third laureate this year, in the field of humanities and social sciences, was prof. Adam Łajtar – awarded “for the interpretation of epigraphic sources, showing the religious and cultural aspects of the functioning of medieval communities inhabiting the Nile Valley”.[10]
Discovery of new minerals (November 2022)
El Ali, also known as Nightfall Is a 15.2-tonne meteorite that was first identified in Somalia in 2020. After two years of studying the 70-gram sample, scientists from the Canadian University of Alberta in Egmont discovered two minerals in it that had not been seen on Earth before.
The discovered minerals were named elalite (in honor of the meteorite and the town near which it was discovered) and elchistantonite (after NASA researcher Linda Elkins-Tanton). The researchers announced the discovery of new chemical compounds on November 21, during the Space Exploration Symposium held at the University of Alberta. It is worth noting that although minerals have not been seen in their natural form on our planet, very similar ones were created synthetically in laboratories in the 1980s. Research on the new compounds will help answer the question of what applications these minerals can find in our world in the future.[11][12]
Breakthrough in thermonuclear fusion research (December 2022)
December 5, 2022 became an important day for the world of science, but also for the history of mankind. On this day, Lawrence Livermore National Laboratory (LLNL) scientists achieved a breakthrough in thermonuclear fusion research, which has been conducted at the National Ignition Facility (NIF). For the first time in history, fusion has produced more energy than was expended to start the reaction. The news of this event was announced on December 13 during a press conference of the LLNL representatives, which was held in the presence of the secretary of the Department of Energy and the head of the US nuclear safety agency.
Thermonuclear fusion consists in combining light atomic nuclei into heavier ones, which is accompanied by the release of significant amounts of energy. The fuel that would be ideally suited to generate energy as a result of this reaction is hydrogen, because we have it in abundance on our planet. However, atomic nuclei repel electrostatic forces, so for the fusion to occur, very specific conditions must be created, namely heating them to millions of degrees and squeezing them to millions of atmospheres (this process is different in stars, where it takes place thanks to quantum tunneling).
Thermonuclear fusion has been attempted many times around the world, but so far the result has been the absorption of more energy than the amount produced. The National Ignition Facility has been working on this phenomenon since the 1950s, but it is technically very difficult. That is why the latest results are such a huge breakthrough and show new possibilities.
The experiment was that the pulse of huge NIF lasers delivered 2.05 megajoules of energy to the hydrogen capsule, while the fusion yielded 3.15 megajoules, or 54% of the excess (over a million joules).
Although a million sounds very exciting, this value is equivalent to a quarter of a kilowatt hour, which is enough energy to boil a kettle of water a dozen times. The vigilant scientific community also notes that while only 2.05 megajoules of energy were supplied to the process itself, more than 322 megajoules of energy was used to power the 192 lasers needed, which is practically a hundred times more than the fusion produced. This is one of the reasons that emphasizes that it takes decades of work by teams of scientists and engineers to find the possibility of using thermonuclear fusion on a larger scale.[13][14]
Ultra-thin solar cells (December 2022)
Much is said about the fact that renewable energy sources are the future of our planet, which can significantly affect the containment of the climate crisis and global warming. That is why scientists are still looking for solutions that will help to use energy from RES even better and easier.
Engineers at the Massachusetts Institute of Technology have created innovative solar cells that can turn any solid surface into a source of energy, and are thinner than a human hair. These cells are glued to a light and very strong fabric, which allows to easily mount them virtually anywhere. According to the scientists, the invention can be very practical in emergencies, when there is no other source of energy in the area, but also when travelling.
The modern cell was made of semiconductor ink with the help of a 3D printer. It is one hundred times lighter than conventional panels, and additionally generates much more energy per kilogram. The solution is still in the testing phase, because there are problems related to the resistance of the panels to environmental factors. However, scientists are working on creating ultra-light containers in which the cells could be enclosed. Researchers believe that ultra-thin cells will be a revolutionary invention for energy generation in the world[15][16].
The strongest material for Earth (December 2022)
Researchers from Lawrence Berkeley National Laboratory (Berkeley Lab) and Oak Ridge National Laboratory conducted research on a new metal alloy that revealed its unusually high ductility (it is malleable and very ductile) and at the same time unprecedented strength (resistant to deformation). This alloy consists of chromium, cobalt and nickel – CrCoNi.
Based on the first tests performed on CrCoNi, it was found that its ductility and strength improve with the cooling of the alloy, even to a temperature of about -196oC. However, the latest research, published in December 2022 in the journal Science[17],
confirmed that it is able to withstand even lower temperatures (-253oC) where liquid helium is present. This is a very interesting phenomenon, because for most other materials the effect is opposite; for example, steel cracks much easier at very low temperatures.
It is worth noting that the CrCoNi alloy belongs to the group of HEA alloys (high entropy alloys). They are distinguished by the fact that they are a mixture of constituent elements in equal parts, rather than with a predominance of one element and a smaller amount of additional elements, as it is the case for most currently used alloys. This has a significant impact on its extraordinary properties.
The unique strength of the CrCoNi alloy at incredibly low temperatures may make it applicable in the future, among others, in objects traversing space.[18]
[1] https://www.nature.com/articles/s41567-021-01463-1
[2] https://www.national-geographic.pl/artykul/ile-wazy-neutrino-naukowcy-w-koncu-zwazyli-najlzejsza-czastke-elementarna-znana-fizyce-220216091750
[3] https://www.iea.org/news/global-co2-emissions-rebounded-to-their-highest-level-in-history-in-2021
[4] https://pubs.acs.org/doi/10.1021/acsnano.2c00955
[5] https://www.nature.com/articles/s41467-022-29624-7
[6] https://naukawpolsce.pl/aktualnosci/news%2C92110%2Cpolacy-stworzyli-magnetyczna-czasteczke-o-wyjatkowych-wlasciwosciach.html
[7] https://www.national-geographic.pl/artykul/nadchodza-baterie-sodowe-tansze-od-litowych-rownie-sprawne-i-bezpieczniejsze-dla-swiata-211207050535
[8] https://www.nobelprize.org/prizes/chemistry/2022/press-release/
[9] https://www.fnp.org.pl/prof-bartosz-grzybowski-laureat-nagrody-fnp-2022/
[10] https://www.fnp.org.pl/znamy-laureatow-nagrod-fnp-2022/
[11] https://www.ualberta.ca/folio/2022/11/new-minerals-discovered-in-massive-meteorite-may-reveal-clues-to-asteroid-formation.html
[12] https://www.national-geographic.pl/artykul/w-tym-meteorycie-odnaleziono-dwa-mineraly-ktorych-nigdy-nie-widziano-na-ziemi-221201050211
[13] https://www.llnl.gov/news/national-ignition-facility-achieves-fusion-ignition
[14] https://oko.press/przelom-w-badaniach-nad-fuzja-termojadrowa
[15] https://news.mit.edu/2022/ultrathin-solar-cells-1209
[16] https://www.national-geographic.pl/artykul/naukowcy-opracowali-ultracienkie-ogniwa-sloneczne-moga-zmienic-kazda-powierzchnie-w-zrodlo-energii-221212125125
[17] https://www.science.org/doi/10.1126/science.abp8070
[18] https://mobirank.pl/2022/12/14/prosty-stop-tworzy-najtwardszy-material-jaki-kiedykolwiek-zarejestrowano/
TOP 10 2021
To show at least a glimpse of these changes, we have prepared a summary of ten interesting discoveries and events of 2021 in the field of chemistry.


Transparent wood (01.21)
Researchers at the University of Maryland have discovered a new technique to make wood transparent. In the past, attempts had been made to make wood transparent by using specialised chemicals to remove lignin. However, the main drawback was that this weakened the wood.
The new method uses an alteration of the lignin. At the beginning of the process, the molecules responsible for giving the wood its colour are removed. Then, a special hydrogen peroxide agent is applied to its surface which is then exposed to UV light (or natural sunlight). After these treatments, the wood takes on a white colour. The wood is then soaked in ethanol for a more thorough cleaning. Finally, the pores are filled with colourless epoxy to make the material smooth and almost perfectly transparent. This gives the wood the qualities of being able to transmit up to 90% of light and the material is 50 times stronger than conventionally rendered transparent material. It is also lighter and, above all, stronger than glass and provides better insulation.[1][2]
This discovery could become a real revolution for the construction industry and completely change the image of buildings in the future. Research is also underway into technologically advanced, transparent wood materials, which will additionally be touch-sensitive and will provide an alternative to various types of displays. Due to their strength corresponding to the characteristics of wood, such displays will prove themselves in harsh environments where glass often fails.[3]

Ink for digital printing on porcelain (03.21)
Ceramic manufacturing methods are distinguished by a long tradition. However, with the development of technology, the time has come for change here too. The digital colouring of ceramic tiles, which may replace the classic glazing method, is to become a breakthrough for this industry. Patterns will be applied with a high resolution printing method, because of this it will be possible to obtain not only various colours, but also various textures, which may be likened to those of fabrics or wood.
The solution has been developed by Italian company Metco, which has created a special, sustainable ink called ECO-INK for digital ceramics. The proposed ink is aqueous, so it does not contain organic solvents, which contributes to reducing both the toxicity and carbon footprint of the product. Additionally, the paint can penetrate the surface of the ceramic tile, thereby eliminating the need for an additional protective layer. This results in a more efficient and sustainable process. Moreover, the surface of the tiles becomes more durable after the application of ECO-INK.
As announced by the manufacturers themselves, this paint is a true revolution for the chemical industry.[4]

Magnetic polymers (03.21)
The magnets we are familiar with are usually found in the form of inflexible and hard metals. These characteristics cause many limitations in the application of magnets. That is why scientists have undertaken the MAGNETO[5]project, which involves creating magnetic materials with mouldable properties.
To achieve this effect, the researchers prepared a powder consisting of shredded magnetic materials that was mixed with various polymers. Advanced 3D printing was used to create a magnet from these components. This made it possible to give them much more complex shapes. The first prototypes produced showed the enormous potential of such materials and the possibility of using them in many fields, from diagnostic tools to touch screens and many others.
The presented composite materials with exceptional magneto-mechanical properties will allow for the introduction of innovative solutions in many areas, such as medicine. Therefore, this represents an important milestone for the development of science and technology.[6]
Newly discovered effects of a natural medicine with one thousand years of history (04.21)
Research has been carried out at the University of Warwick into an ‘antibiotic’ vegetable paste whose recipe is 1,000 years old. It is called ‘vision repairing ointment’, and it was discovered in the Old English medical manual Medicanale Anglicum, written in the 9th century. The ointment, which contains onions, garlic (or leek – the scientists had trouble translating the name correctly), cow bile and wine, has extremely potent antiseptic properties. It has been shown to be effective against certain strains of bacteria that have become resistant to modern drugs.
Even initial testing has proven the concoction’s effectiveness in treating Staphylococcus aureus. However, recent research has been extended to other strains and the results have been presented in the form of a scientific publication.[7] Experiments have shown that this natural medicine can be a powerful weapon against bacteria called biofilms. This is one of the most dangerous types of bacteria, among which, we can find strains causing, for example, sepsis, but also other serious infections. It is also hoped that this recipe will help to treat foot infections in diabetics, for example, which at the moment often result in amputation.
The example of the paste described above draws attention to the clash between natural medicine and modern pharmaceuticals. It leads one to draw new conclusions and inspires hope for the treatment of diseases that cause suffering to many people.[8]
Vanilla flavouring based on plastic (06.21)
The problem of the disposal of objects made from plastic is one of the biggest challenges of the present time. The whole world is struggling to develop effective methods to reduce the amount of pollution that devastates our environment. One of the most interesting solutions turned out to originate from scientists at the University of Edinburgh, who transformed plastic bottles into vanilla flavouring. The research involved mutating the enzymes responsible for the decomposition of polyethylene terephthalate (the polymer from which the bottles are made). The decomposition reaction produced terephthalic acid (TA), which was then converted into vanillin. This compound carries most of the taste and smell of vanilla and is often used in the food, pharmaceutical and cosmetics industries.
According to The Guardian magazine, which published extracts of an interview with Joanna Sandler of the University of Edinburgh, who led the research project, 85% of vanillin is currently synthesised from chemicals derived from fossil fuels.[9] However, the demand for vanillin continues to rise. Therefore, this is an important discovery both because of the increase in demand, but more importantly for the sake of a solution with environmental benefits.[10]
Plastic-eating yeasts to save the planet (09.2021)
The environmental pollution caused by plastics is one of the biggest environmental disasters. Microparticles of plastics, which have a diameter of less than 5 millimetres, pose a particular threat. They can be found in water bodies, but also accumulate in living organisms such as fish, plankton and the human body.
This problem has been addressed by the research team of Dr Piotr Biniarz from the Wrocław University of Environmental and Life Sciences. Their research consists of finding microorganisms that naturally decompose plastics due to the enzymes that they possess. However, as this process is usually inefficient, it is planned to clone their enzymes into fast-growing yeasts (Yarrowia lipolytica). These organisms will not only be able to produce enzymes more efficiently, but also to grow on municipal wastewater or waste so that micropollutants can be removed directly from it.[[11]

Nobel Prize 2021 (10.2021)
This year’s Nobel Prize in Chemistry was awarded to David MacMillan and Benjamin List ‘for the development of asymmetric organic catalysis’. Organocatalysis is a unique tool for building molecules. Until this discovery, it was assumed that there were only two types of catalysts, or substances that speed up the course of chemical reactions. These are enzymes and metals. However, scientists have recently demonstrated the existence of asymmetric organic catalysis, which uses small organic molecules.
Organic catalysts are characterised by a stable skeleton of carbon atoms, to which chemical groups with a higher activity can attach. They may contain elements such as sulphur, nitrogen, oxygen or phosphorus. They are much smaller than enzymes, which facilitates their production. These features make the catalysts more environmentally friendly, but also relatively inexpensive to produce.
Asymmetric organic catalysis has been developing since 2000, and David MacMillan and Benjamin List are the clear leaders in the field. Their discovery has shed new light on many conventional industrial processes and shown that organic catalysis may be used in many chemical reactions. It is highly efficient and can support the manufacture of almost anything from modern pharmaceuticals to the molecules responsible for capturing light in photovoltaic cells. This discovery has definitely revolutionised the world of science and technology.[12][13]

The material that feels (12.21)
A research group consisting of scientists from Chicago and Missouri set out to design a material that is sensitive to sensing surrounding stimuli and adapting to them.
As it has properties that are not present in naturally occurring materials, it belongs to the group of so-called metamaterials. It is made from piezoelectric elements that are controlled by electrical circuits. It may be used to form a specialised circuit that processes information. In addition, electrical energy enables it to move and change form. These elements allow it to sense external stimuli and adapt to them. As the creators themselves say, this material is able to make decisions without human interference.
Such a metamaterial could work very well in aviation, the space industry, medicine and in many other areas.[14][15]

Eco-friendly plastic from salmon seed (12.21)
Plastics were supposed to constitute a revolution among available materials. However, despite their many advantages, they have also become one of the main problems threatening our planet. This is why research into greener alternatives is continuing.
Chinese scientists have developed a unique plastic-like material, one of the main components of which is salmon seed. This was accomplished by combining two strands of salmon DNA with a chemical derived from vegetable oil. The result is a spongy, gel-like substance – a hydrogel. The resulting hydrogel is freeze-dried and moisture is removed from it, thereby allowing it to be moulded into different shapes.
The production of this bioplastic can emit up to 97% less CO2 than the production of traditional polystyrene plastics. Additionally, it will be recyclable using DNA digesting enzymes. Ultimately, it can also be immersed in water so that it becomes a hydrogel again.
These types of bioplastics represent an opportunity for the future of the plastics industry and to reduce the pollution on our planet.[16]
Graphene-based lubricant (12.21)
Italian researchers have developed a novel graphene-based lubricant that can be used in cars and motorbikes. In particular, the addition of graphene has ensured the greater stability of oil which, in addition, helps to reduce the friction between engine parts. These beneficial properties make the parts heat up and also wear out less rapidly. Graphene has the potential to become an alternative to traditionally used oil. This will make oil less toxic to the environment and also make it easier to dispose of or recycle. The lubricant has already undergone its first tests, in which it performed to a promising level. Therefore, further research is underway to bring this graphene innovation to commercial applications.[17]
[1] https://dzienniknaukowy.pl/nowe-technologie/nowy-sposob-na-przezroczyste-drewno-ktore-mogloby-zastapic-szklo-w-naszych-oknach
[2] https://www.science.org/doi/10.1126/sciadv.abd7342
[3] https://cordis.europa.eu/article/id/429178-translucent-touch-sensitive-wood-biomaterials-revolutionising-wood-in-construction-and-beyond/pl
[4] https://cordis.europa.eu/article/id/430550-an-innovative-sustainable-ink-for-printing-digital-porcelain/pl
[5] http://www.kostasdanas.com/erc-magneto/
[6] https://cordis.europa.eu/article/id/434341-magnetic-polymers-set-to-be-a-material-of-the-future/pl
[7] https://www.nature.com/articles/s41598-020-69273-8#Sec9
[8] https://www.national-geographic.pl/artykul/sredniowieczna-mikstura-odtworzona-w-laboratorium-niszczy-lekooporne-bakterie
[9] https://www.theguardian.com/environment/2021/jun/15/scientists-convert-used-plastic-bottles-into-vanilla-flavouring
[10] https://forsal.pl/biznes/ekologia/artykuly/8191441,naukowcy-przetwarzaja-plastikowe-butelki-na-aromat-waniliowy.html
[11] https://perspektywy.pl/portal/index.php?option=com_content&view=article&id=6413:drozdze-zjadajace-plastiki-naukowcy-z-upwr-pomoga-planecie&catid=24&Itemid=119
[12] https://www.focus.pl/artykul/nagroda-nobla-2021-nobel-z-chemii-za-genialne-narzedzie-do-budowania-czasteczek-211006123039
[13] https://www.nobelprize.org/prizes/chemistry/2021/press-release//p>
[14] https://www.chip.pl/2021/12/material-reaguje-na-bodzce-technologie-stealth/
[15] https://www.nature.com/articles/s41467-021-26034-z
[16] https://www.national-geographic.pl/artykul/naukowcy-stworzyli-ekologiczny-plastik-z-nasienia-lososia
[17] https://cordis.europa.eu/article/id/429711-graphene-based-lubricant-boosts-engine-performance/pl
TOP 10 2020 – What surprised us in 2020?
We have a difficult year behind us, which we will mainly associate with the COVID-19 pandemic. Fortunately, science has been beyond that and many exceptional discoveries have also been made during this time.
Let us therefore summarise some of the most important events for the world of chemistry, which will have an impact on our future and further development of science.


A TELESCOPE THAT BRINGS US CLOSER TO THE SUN (January 2020)
A telescope that made it possible to capture extremely detailed pictures of the Sun has been built in Hawaii by National Science Foundation (NSF), a US government agency. It is the world’s largest telescope and has a 4-metre solar mirror. The pictures it takes has created a new era in the study of the Sun. It will enable weather forecasters to predict geomagnetic storms more accurately and better understand what affects cosmic weather. [1]
A YEAR MARKED BY THE COVID-19 PANDEMIC (March 2020)
Even though the first COVID-19 cases was observed in November 2019, the World Health Organisation labelled it as a pandemic on 11 March 2020. The disease caused by the SARS-CoV-2 virus shook the whole world. New recommendations and orders have changed our everyday reality. An important role has been played by chemicals such as disinfectants, which proved to be an important weapon in the fight against the spread of the disease. The chemical industry also took an important role in the medical and pharmaceutical sectors by supporting doctors in their fight against the disease.
PLASTIC-EATING BACTERIA (April 2020)
On 8 April 2020, Nature published an article proving the existence of bacteria with enzymes that are capable of breaking down plastics and turning them into simple elements. During digestion, strain 201-F6 b of Ideonella sakaiensis makes it possible to recover material that can be used again in the synthesis and production of plastics of the same quality as that obtained through petrochemical processes. This method is being slowly implemented in industry, and in a few years we should be able to buy recycled bottles manufactured using this method. [2]

A WAY TO CUT 2D MATERIALS (14 July 2020)
Scientists have developed a very precise technology which makes it possible to carve small holes in atom-sized particles. The aim is to support the production of photonic and electronic nanodevices. Research describes a thermomechanical technique which makes it possible to cut 2D materials using a heated scanning nanotip. This method makes it possible to make arbitrarily shaped cuts with a resolution of 20 nm in monolayer 2D materials. [3]
METAL-EATING BACTERIA (15 July 2020)
For over 100 years scientists have suspected the existence of metal-eating bacteria. However, they could not prove it until now. The discovery was made by microbiologists from Caltech (California Institute of Technology). Dr. Jared Leadbetter was conducting research based on manganese. When he finished, he placed a glass jar he had been using in the sink to soak. By coincidence and because he had to leave the campus, the jar was left in the water for several months. When Leadbetter returned, he discovered that the vessel was coated with a dark residue, which turned out to be oxidised manganese generated by the bacteria living in tap water. Extensive research has shown that the bacteria can use manganese for chemosynthesis. It is the first known case in which bacteria use manganese as a source of energy. It is a revolutionary step for science, one which has largely contributed to our understanding of natural elemental cycles. [4]

ALMOST INVISIBLE FISH (17 July 2020)
The unique fish in question are real masters of camouflage. Their black exterior absorbs 99.95 percent of all photons. These fish literally soak up all the light, so even under a strong spotlight we can only see their silhouettes against dark water. Karen Osborn, a research zoologist at the Smithsonian’s National Museum of Natural History, and her team discovered 16 species of fish which look as if they were covered with Vantablack, the darkest material known to humans, which absorbs 99.96 percent of light. [5]

THE NOBEL PRIZE IN CHEMISTRY (October 2020)
Emmanuelle Charpentier and Jennifer A. Doudna were awarded the Nobel Prize for the development of a method for genome editing. They discovered precise “genetic scissors” which could, for example, make it possible to develop new cancer therapies. The method was discovered in 2012 and was a scientific breakthrough. [6]

A RECORD-SETTING TIME MEASUREMENT: ZEPTOSECONDS (19 October 2020)
Scientists managed to measure the shortest time unit, known as the zeptosecond. It was measured during the observation of a light particle crossing a hydrogen molecule. It took 247 zs (zeptoseconds). It was decided that one zeptosecond was 10-21 of a second.
Measurements were made by a team of physicists lead by Professor Reinhard Dörner of Goethe University in Frankfurt am Main, Germany. [7]

THE POLISH NOBEL PRIZE (4 November 2020)
Other awards granted this year included those of the Foundation for Polish Science (also referred to as the Polish Nobel Prize). In the field of chemistry, the prize was awarded to Professor Ewa Górecka of the University of Warsaw “for obtaining liquid crystal materials with a chiral structure made of non-chiral molecules.” [8]

DIAMONDS MADE IN MINUTES (20 November 2020)
Scientists of the Australian National University (ANU) managed to create a diamond just by applying high pressures and without raising the ambient temperature. They obtained two types of diamond. One was a typical stone, which could be used on a ring after cutting. The second type was called lonsdaleite, a form which is found in nature after a meteorite hits the Earth. The possibility of creating a diamond so quickly and at room temperature opens up multiple possibilities, including for industry. [9]
[1] https://edition.cnn.com/2020/01/29/world/sun-image-inouye-telescope-scn/index.html
[2] https://www.nature.com/articles/s41586-020-2149-4
[3] https://onlinelibrary.wiley.com/doi/full/10.1002/adma.202001232
[4] https://www.nature.com/articles/s41586-020-2468-5.epdf
[5] https://www.scimex.org/newsfeed/ultra-black-fish-are-practically-invisible
[6] https://www.nobelprize.org/prizes/chemistry/2020/press-release/
[7] https://aktuelles.uni-frankfurt.de/forschung/physik-zepto-sekunden-neuer-weltrekord-in-kurzzeit-messung/
[8] https://www.fnp.org.pl/nagrody-fundacji-na-rzecz-nauki-polskiej-2020-przyznane/
[9] https://edition.cnn.com/2020/11/19/world/diamonds-room-temperature-scli-intl-scn/index.html
What scientific discoveries did 2019 bring?
We have a very special time behind us, because last year was the 150th anniversary of the discovery of the Periodic table by Dmitri Mendeleev. To honour this milestone in chemistry, the General Assembly of the United Nations (UN) and UNESCO declared 2019 the “International Year of the Periodic Table of Chemical Elements (IYPT2019)”. In connection with this event, have a look at our Facebook fanpage, where we have organized a unique contest on the knowledge of the elements and periodic table. Apart from a special anniversary, this year was full of new discoveries. We have selected the 10 most interesting ones, among which there are, for example, spectacular results of research on the new state of matter, the method of using sunlight to produce fuels or creating cyclocarbon. Below is a calendar of the 10 most interesting chemical discoveries and events of 2019.
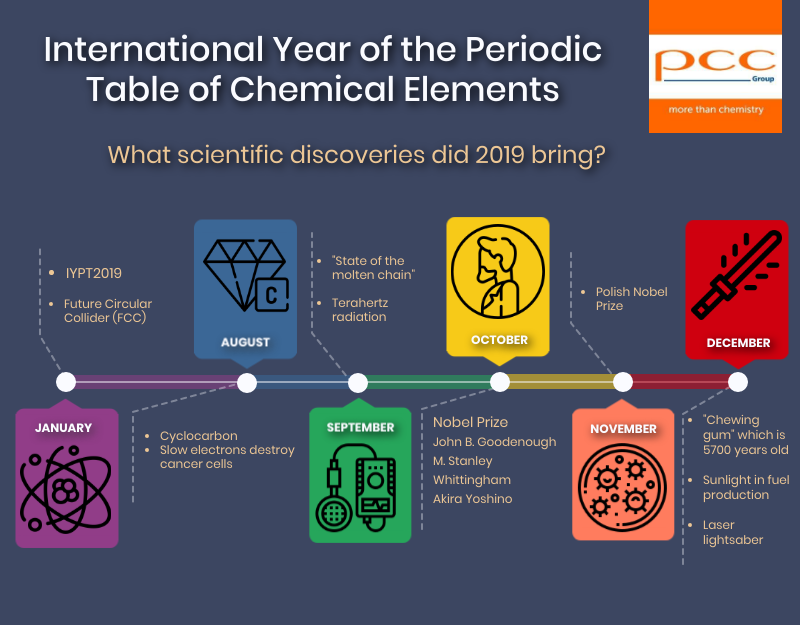

BUILDING A NEW HADRON COLLIDER Future Circular Collider (FCC)
The FCC is to be four times bigger and many times more powerful than the Large Hadron Collider (LHC). The accelerators allow to examine the elements created by the collision of streams of accelerated elementary particles. Accelerator with a larger size and greater power may allow us to discover yet unknown forms of matter and to investigate more thoroughly the already known ones.[1]

CYCLOCARBON A NEW VARIETY OF CARBON
Scientists from the University of Oxford and IBM Research in Zurich, in a publication in “Science” magazine, presented how to produce a ring made of 18 carbon atoms. This relationship was created by an innovative method of manipulating single atoms. One of the discoverers of cyclocarbon was a Pole Dr Przemysław Gaweł from the University of Oxford.[2]
SLOW ELECTRONS DESTROY CANCER CELLS
Scientists from the Vienna University of Technology discovered that the previously observed effect of destroying cancer cells using slow electrons is possible. By using the Coulomb’s interatomic decomposition, the ion can transfer additional energy to the surrounding atoms. As a result, a huge number of electrons are released, with enough energy to cause DNA damage to cancer cells.[3]
NEW STATE OF MATTER
A team of scientists from the University of Edinburgh performed computer simulations to further investigate the so-called “state of the molten chain“. The tests were carried out on 20 000 potassium atoms subjected to a pressure of 20 000 to 40 000 atmospheres and a temperature of 126 to 526 degrees Celsius. The results showed that the created structures represent a new state in which two interconnected lattice structures are formed. The observation is that chains dissolve into a liquid while at the same time the remaining potassium crystals are in a solid form.[4]

NEW TERAHERTZ RADIATION SOURCES
Scientists from the CENTERA research agenda, together with research teams from France, Germany and Russia, have made a discovery that may lead to the construction of new sources of forgotten terahertz radiation. It would be tunable with a magnetic field. The results of these studies are described in Nature Photonics.[5]

NOBEL PRIZE IN CHEMISTRY
John B. Goodenough, M. Stanley Whittingham and Akira Yoshino were awarded for the development of light and capacious lithium-ion batteries. This invention is commonly known as lithium-ion batteries. Their creation revolutionized the world and, as the members of the Nobel Committee pointed out, “they laid the foundations of a wireless, fossil fuel-free society“. [6]

POLISH NOBEL
The winner of the Foundation for Polish Science Award (the so-called Polish Nobel Prize) is Professor Marcin Drąg from the Faculty of Chemistry of the Wrocław University of Technology. Professor was appreciated “for developing a new technological platform for obtaining biologically active compounds, especially proteolytic enzyme inhibitors.”[7]

DNA in a prehistoric “chewing gum”
Scientists from the University of Copenhagen report in “Nature Communications” about finding a DNA fragment of a prehistoric inhabitant of Scandinavia in a piece of birch tar she chewed. Based on this discovery, the complete female genome was reconstructed. The artefact dates back to 5700 years.[8]

SUNLIGHT FOR FUEL PRODUCTION
Researchers at Nanyang Technological University of Singapore (NTU Singapore) have discovered a method that can transform plastic waste into chemicals by using sunlight. A team of scientists conducted research on a mixture of plastics with their catalyst in a solvent, which allows the use of light energy. As a result, dissolved plastics were transformed into formic acid. This acid is used in fuel cells to produce electricity. This discovery is aimed at developing sustainable methods of using sunlight to produce fuels and other chemical products. [9]

LASER LIGHTSABER AT OUR FINGERTIPS
Aleksandra Fliszkiewicz, a student of the Warsaw University of Technology, developed a light swordas part of her engineering work, inspired by the 8th part of “Star Wars”. It was created using a green laser and a lens developed by Polish scientists, the so-called “light sword”, which focuses the light into a section. The lens, the geometry of which was developed in 1990 at the Warsaw University of Technology, is now also supposed to bring new solutions in ophthalmology, such as the creation of intraocular implants for people after cataract surgery, which are being clinically tested.[10]
[1] https://www.bbc.com/news/science-environment-46862486?ns_campaign=bbcnews&ns_mchannel=social&ns_source=facebook&ocid=socialflow_facebook&fbclid=IwAR3th4hAdlz5ww5JJdTnn5b3MJv5PxVP8inCpYaNlRBjA3FaCq-1Y5SPzcs
[2] https://science.sciencemag.org/content/365/6459/1299
[3] https://www.sciencedaily.com/releases/2019/08/190822101429.htm
[4] https://www.nationalgeographic.com/science/2019/04/new-phase-matter-confirmed-solid-and-liquid-same-time-potassium-physics/
[5] https://www.fnp.org.pl/w-poszukiwaniu-nowych-zrodel-promieniowania-terahercowego/
[6] https://www.nobelprize.org/prizes/chemistry/2019/press-release/
[7] https://www.fnp.org.pl/laureci-nagrody-fnp/
[8] https://healthsciences.ku.dk/newsfaculty-news/2019/12/ancient-chewing-gum-yields-insights-into-people-and-bacteria-of-the-past/
[9] https://www.sciencedaily.com/releases/2019/12/191211100331.htm
[10] http://naukawpolsce.pap.pl/aktualnosci/news%2C80037%2Cna-politechnice-warszawskiej-powstal-laserowy-miecz-swietlny.html































































































































