The agrochemical industry focuses its efforts on developing effective pesticide formulations that support efficient agricultural production. A key element of these tasks is the development of functional additives.

For pesticide formulations to work effectively, their ingredients must be properly composed and compatible. In addition to active substances, inert ingredients such as carriers, solvents and adjuvants are important components of pesticide formulations. Although the latter do not have a direct pesticidal effect, they are added to the formulation to improve its effectiveness.
Many pesticide formulations available on the market come in the form of emulsifiable concentrates (EC) and concentrated water-in-oil emulsions (EW), which are thermodynamically unstable systems. During storage, they may undergo adverse phenomena such as coalescence, flocculation, phase separation or crystallisation of active ingredients. Emulsification efficiency may also deteriorate, leading to the precipitation of cream or oil in ready-to-use spray liquids. Most active substances in pesticides are non-polar compounds that do not dissolve in water but dissolve well in organic solvents. In such cases, the selection of solvents and emulsifiers is fairly straightforward and it is easy to avoid the aforementioned defects. It is much more difficult to achieve this for substances that are partially soluble in water (e.g. tebuconazole) and require specific solvents, as in such cases it is difficult to achieve full solubility of the substance in the formulation over the entire storage temperature range and, in addition, crystals easily precipitate in the finished spray liquid. Therefore, in addition to special solvents for such difficult cases, crystallisation inhibitors are often used, which further complicates the system. Different combinations of solvents and active ingredients require appropriate emulsifiers [1, 2, 3].
What is an emulsifier?
Emulsifiers are chemical compounds that facilitate the mixing of immiscible liquid-liquid systems. They are usually precisely composed mixtures of various surfactants, rather than single substances.
The properties of an emulsifier are determined by its molecular structure. Based on their structure, emulsifiers can be divided into ionic and non-ionic. Ionic emulsifiers (mostly anionic sulphates and phosphate esters) usually consist of a hydrophilic “head” with a charge and a hydrophobic “tail”. This dual structure is called amphiphilic and is key to the functioning of emulsifiers. Individual molecules concentrate at the boundary between two immiscible phases with different polarities. The “head” of the emulsifier interacts with the hydrophilic phase, while the “tail” interacts with the hydrophobic phase. This dual affinity allows emulsifiers to locate themselves at the phase boundary, creating a barrier that prevents the coalescence of emulsion droplets and additionally stabilises the emulsion electrostatically. In ionic emulsifier molecules, there is often a polyoxyethylene glycol chain between the head and the tail, the presence of which further increases the effectiveness of the emulsifiers [4, 5].
Non-ionic emulsifiers, on the other hand, consist only of fragments that do not have electrical charges. Their role is also very important, because when ionic emulsifier molecules repel each other and are unable to form a tight barrier at the phase boundary, non-ionic emulsifiers come to the rescue, easily filling the gaps between them [4, 5].
Both ionic and non-ionic emulsifiers do not always have to have the proverbial head-and-tail structure; they can also consist of hydrophobic parts on the sides and a hydrophilic part in the centre, or vice versa, which allows for the creation of very stable microemulsions. They can also have a branched hydrophobic part and a linear hydrophilic part, or even consist of several linear hydrophobic and hydrophilic parts joined together. Such complex structures stabilise emulsions very well in a steric manner, and their complexity is the result of numerous syntheses and application tests [4, 5].
Emulsifiers perform many different functions:
- They reduce the interfacial free energy.
- They reduce surface tension.
- They form a layer or barrier around the droplets of immiscible phases.
- They facilitate emulsification.
- They cause electrostatic and steric stabilisation [4, 5].
The importance of emulsifiers in pesticide formulations
Emulsifiers, as components of pesticide formulations, do not control pests themselves, but they play a significant role in them. Their importance can be summarised in several key points [6]:
– Emulsion stabilisation – emulsion stability is the ability to resist particle aggregation, which would lead to phase separation. Emulsions are thermodynamically unstable systems with high free energy. Low emulsion stability is a common problem faced by farmers. If the emulsifier is not selected appropriately, the individual phases of the emulsion quickly separate.
– Improved application properties – emulsifiers ensure uniform dispersion of the active substance throughout the entire volume of the preparation. After spraying, a well-prepared formulation ensures even and precise distribution of the pesticide on the crop, which is essential for effective pest control.
– Increased spraying efficiency – emulsifiers also act as built-in adjuvants. Thanks to their small and mobile molecules, they quickly reach the boundaries of the freshly formed droplets during spraying and regulate their size during secondary droplet division. This prevents the formation of droplets that are too large, which quickly fall to the ground, or too small, which are easily blown away by the wind. The same emulsifier molecules, by reducing surface tension, prevent droplets from bouncing off and running off leaves upon contact. Emulsifiers based on vegetable oils or their acids, on the other hand, also loosen the cuticular wax very well, which facilitates the absorption of systemic pesticides.

Emulsifier – the key to a stable emulsion
The selection of an emulsifier for a pesticide formulation is an important issue that has a direct impact on the effectiveness of spraying.
Ease of use is equally important. The low viscosity and melting point of the emulsifier allow the heating stage to be bypassed and enable it to be easily pumped through the system. During storage of the finished formulation, the emulsifier must not cause decomposition of the active ingredients, flocculation or precipitation; on the contrary, it must stabilise the formulation. After dilution with water, it should reduce surface tension, emulsify the pesticide formulation properly, and ensure adequate coverage and penetration of the leaves by the spray. Importantly, the emulsifier must be inert and not interact chemically with any of the other ingredients in the formulation [6].
The effectiveness of spraying depends on whether the pesticides meet specific requirements. Therefore, it is important to consider the structure of the emulsifier, including functional groups, their number, hydrophilic-lipophilic balance (HLB), compatibility with solvents, etc. This often requires numerous laboratory trials, followed by testing under real-world conditions [6, 7].
The hydrophilic-lipophilic balance (HLB) of an emulsifier is an important factor when selecting it for use in a pesticide formulation. The most well-known and widely used HLB scale was described by Griffin in 1954. The scale ranges from 1 to 20, where 1 represents the highest hydrophobicity and 20 the highest hydrophilicity. The HLB of an emulsifier is calculated according to this scale using the following formula [6, 7]:
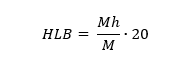
Where:
Mh – molecular weight of the hydrophilic part of the molecule
M – molecular weight of the entire molecule
Pesticide formulations typically use emulsifiers with HLB values between 7 and 17, and in most cases, both a low HLB emulsifier and a high HLB emulsifier should be used in a single formulation, as it has been observed that such combinations produce more stable emulsions than the use of a single emulsifier with a specific HLB value. Another advantage of this solution is its flexibility, which allows the HLB of the entire system to be easily increased or decreased by changing the ratio of the two emulsifiers [6, 7].
Although Griffin’s method is very simple for calculating HLB, it cannot be overlooked that it is very general – it only takes into account the ratio of the hydrophilic and hydrophobic parts. This approach works well for simple molecules with a non-ionic structure. However, empirical methods are better suited for determining the HLB of emulsifiers with complex and often ionic structures, the most popular of which is the Davies method. In 1957, Davies analysed a large number of emulsifiers with different structures and noticed that each functional group has a different effect on emulsifying properties. Therefore, he proposed the following equation for calculating HLB [6, 8]:
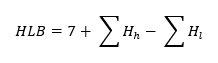
Where:
Hh – HLB values of hydrophilic groups
Hl – HLB values of hydrophobic groups
The table below shows several examples of functional groups and their HLB values:
Table 1. Example values of functional groups according to Davies.
| Functional group | HLB value (increment) | Character |
| –CH₂–, –CH₃ | 0.475 | lipophilic |
| –OH | 1.9 | hydrophilic |
| –COOH | 2.1 | hydrophilic |
| –O– (ether) | 1.3 | hydrophilic |
| –COO– (anion) | 2.1 | hydrophilic |
| –COO–R (ester) | 2.4 | moderately hydrophilic |
| –CONH₂ | 1.9 | hydrophilic |
| –NH₂ | 1.9 | hydrophilic |
| –SO₄⁻ Na⁺ | 38.7 | strongly hydrophilic |
| –PO₄²⁻ (phosphate) | 21.0 | very strongly hydrophilic |
This approach allows for a much more accurate estimation of the HLB of an emulsifier. There are several other empirical methods for determining HLB that use the following for calculations: critical micelle concentration, cloud points, emulsifying properties and foaming properties. Nuclear magnetic resonance ( ) spectroscopy is also very helpful in determining HLB, as it allows the structure of the emulsifier to be determined and HLB to be calculated on the basis of the spectrum obtained [6, 8].

PCC Exol as a supplier of emulsifiers for plant protection product formulations
PCC Exol meets the expectations of pesticide formulation manufacturers by offering a wide range of emulsifiers with different chemical structures and associated application properties.
In emulsifiable pesticide formulations or oil adjuvants, many groups of chemicals are used as emulsifiers. The most popular are alkoxylated alcohols (ROKANOL product group) or fatty acids (ROKAcet product group), which have a linear or branched lipophilic part and a linear hydrophilic part. Such alkoxylates are often additionally sulphated (SULFOROKAnol product group) or phosphorylated (EXOfos product group) and neutralised to appropriate salts in order to obtain electrosteric emulsifiers with an ionic structure. Alkoxylated vegetable oils are also often used as emulsifiers, and are typically characterised by a branched structure. Particularly noteworthy are castor oil ethoxylates (ROKAcet series R), which are widely used in EC, EW and OD formulations, where, thanks to their branched structure, they additionally stabilise the oil suspension against sedimentation [9].
Amines (ROKAmin product group) can also be used as emulsifiers. Amines usually have a non-ionic structure and their use increases the pH of the system. On the other hand, if such an amine undergoes quaternisation and neutralisation, it becomes a cationic surfactant and can be used in systems requiring cationic surfactants [9].
Sorbitol derivatives, i.e. sugar alcohol with 6 hydroxyl groups in an open structure or 4 in a closed structure, are also very important as emulsifiers. Sorbitol and fatty acid esters (sorbitans – ROKwin product group) are known to be used as low HLB emulsifiers, and sorbitol ethoxylates (polysorbates – ROKwinol product group) are used as high HLB emulsifiers. In addition to their emulsifying properties, sorbitol derivatives also have good dispersing and stabilising properties, which makes them valuable as non-aqueous dispersants in OD oil suspensions. Derivatives of other sugars are also known: glucose – alkyl polyglycosides, sucrose esters, mannose or lactose, which can also be used as emulsifiers [9].
The last group worth mentioning are EO/PO or even BO block copolymers (ROKAmer product group), which, depending on the content of individual meres, have specific HLB values and particle sizes. Here, too, depending on the starter and the order of attachment, it is possible to obtain not only linear but also branched structures, which may have a block structure and consist of alternating hydrophilic and hydrophobic blocks. Random copolymers do not work well as emulsifiers due to their non-dual structure [9].
Special cases are CS formulations, where during in situ polymerisation a homogeneous emulsion with specific micelle sizes must be ensured. Therefore, we recommend products such as SULFOROKAnol L227/1, SULFOROKAnol L430/1, SULFOROKAnol TSP95, EXOfos PB 136 and EXOfos PB 139. It should be noted that sulphates usually allow smaller capsules to be obtained than phosphoric esters, but they are more sensitive to the presence of ions and pH fluctuations, which is why a combination of sulphate with a small addition of phosphoric ester is often used to stabilise the system. Non-ionic emulsifiers should also be added, and here we recommend: ROKAnol TSP16, ROKAnol L30/65, ROKAnol IT40/70, ROKAnol UD28/70 and ROKAnol UD40/70. As protective colloids, we recommend polyvinyl alcohol PVA, polyvinylpyrrolidone PVP, hydroxymethylcellulose HEC or carboxymethylcellulose CMC, but we do not offer these products [9].
In summary: The choice of emulsifiers is very wide, and new molecules are constantly being developed to meet the needs of manufacturers of emulsifiable pesticide formulations. Emulsifiers based on natural substances, which are safe for consumers and environmentally neutral, are becoming particularly important. This trend is gaining momentum and is linked to the growing environmental awareness of manufacturers, who are actively seeking to reduce the use of toxic and harmful compounds and are trying to replace petroleum-based raw materials with those of natural origin.
- [1] Pacheco, R., Attard, T., Calvert, M., & others. Green solvent selection for emulsifiable concentrate agrochemical formulations. Organic Process Research and Development, 2023.
- [2] Chasin, D. G. Pesticide concentrated emulsion formulations. ASTM International 1987.
- [3] Lewis, K.A., Tzilivakis, J., Warner, D. and Green, A. An international database for pesticide risk assessments and management. Human and Ecological Risk Assessment: An International Journal, 22(4), 1050-1064, 2016.
- [4] Tadros, T. F. Emulsion science and technology. Wiley-VCH. 2013.
- [5] Stauffer, C. E. Emulgatory. Warszawa: WNT. 2001.
- [6] Rosen, Milton J., and Joy T. Kunjappu. Applied Surfactants: Principles and Applications. Weinheim: Wiley‑VCH, 2012.
- [7] Griffin, W. C. Calculation of HLB values of non ionic surfactants. Journal of the Society of Cosmetic Chemists, 5, 259–268, 1954.
- [8] Davies, J. T. A quantitative kinetic theory of emulsion type. I. Physical chemistry of the emulsifying agent. In Gas/Liquid and Liquid/Liquid Interface: Proceedings of the International Congress of Surface Activity (pp. 426–438). 1957.
- [9] PCC Group, Agrochemicals – Markets and applications, 2026. https://www.products.pcc.eu/en/products/markets-and-applications/agrochemicals/






























































































































