The production of sodium hydroxide on an industrial scale can take place in several ways. Until the 1990s, the most popular production method was the mercury method, which has now been replaced by a more advantageous membrane method.

“The membrane method is the most promising and the fastest growing technique for the production of chlorine and alkali, which will undoubtedly replace the other techniques (diaphragm and mercury) over time”
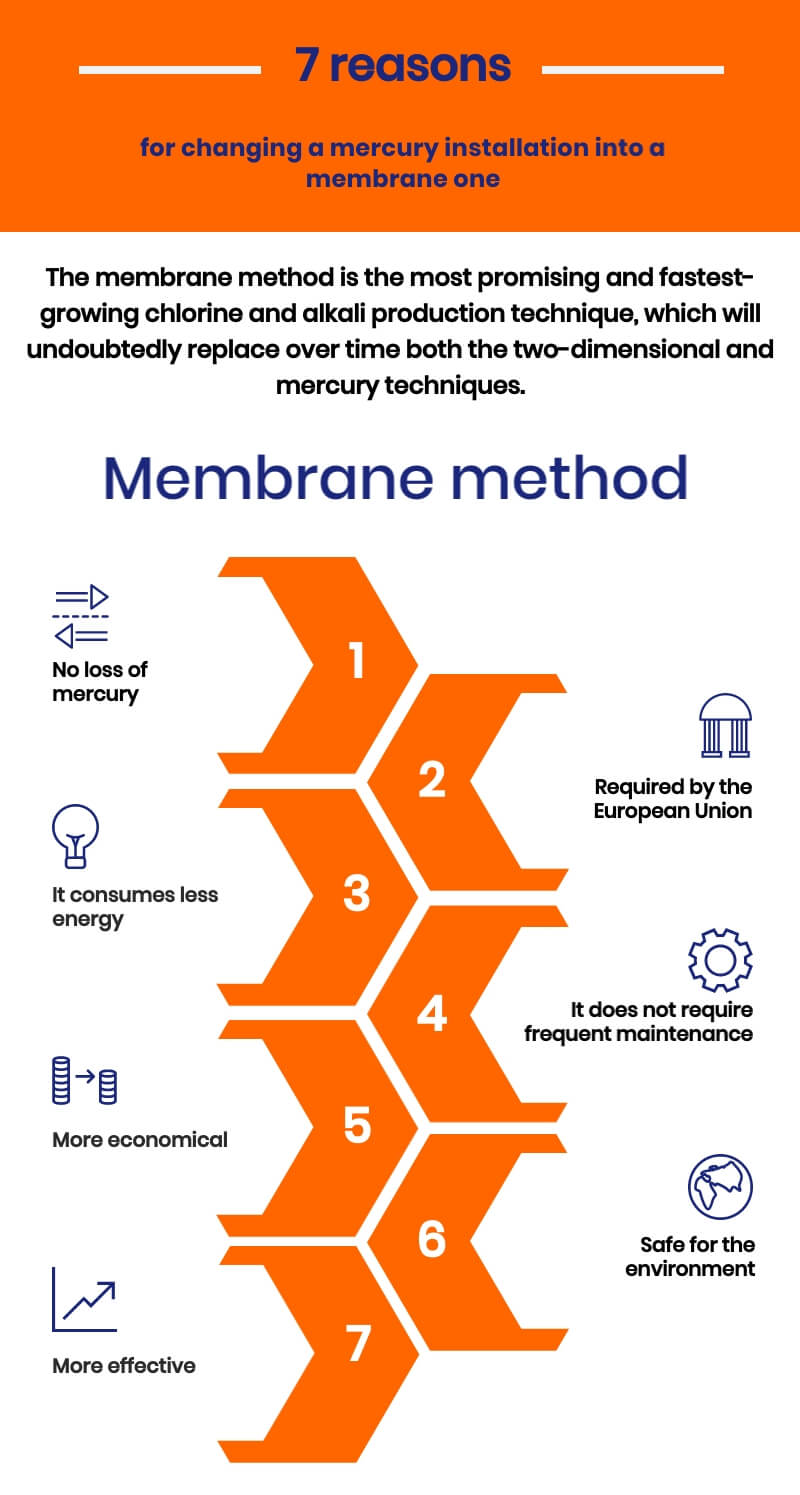
Modern membrane technology is better than mercury because:
- it prevents mercury from entering the natural environment,
- it is more economical (less energy consumption),
- membrane installation does not require as much maintenance as mercury.
The membrane method replaces the mercury method
The mercury process has been widely used in Europe for over 100 years. The beginning of the development of membrane technology, or ion exchange membranes to be exact, dates back to the 1970s. The large-scale membrane process began to be used for the production of sodium hydroxide in 1987. Since then, the popularity of membrane electrolysis has slowly increased, thanks to which more and more chemical plants have transformed mercury into membrane systems. The conversion of mercury technology to membrane technology, however, has occurred very slowly in Europe, mainly due to the high costs of modernisation. As recently as in 2001, 55% of the chlorine-alkaline production capacities installed on our continent used mercury technology, and only 20% of them were made of membrane technology. Only the introduced changes in legal regulations has forced producers to move away from mercury technology. In accordance with the provisions of the Industrial Emissions Directive, effective since 2013, 2017 was the last year in which the mercury method in the production of chloroalkalines, including sodium hydroxide (read about sodium hydroxide suppliers), could be used in EU countries.
The main reasons for the complete withdrawal of mercury electrolysis from the EU are:
- legal requirements,
- economic reasons – high production costs,
- increase in the demand for soda lye,
- bad technical condition of the installation.
The most important reason for changing the mercury plant into a membrane system is the costs and conditions of wear and maintenance of production systems, as well as the condition of the environment and human health. In the case of PCC Rokita, compliance with new legal standards began in 2010, when the first membrane electrolysis plant was commissioned in Brzeg Dolny. The second part was launched in 2015 with the final closure of the plant using the mercury method.
What is membrane electrolysis?
In the production of sodium hydroxide by the membrane method, both electrodes (titanium anode and steel or nickel cathode) separate the membrane which conducts the hydrated ions. In the anode space chloride ions from the brine are oxidised to gaseous chlorine, while sodium ions migrate through the membrane to the cathode space through which sodium hydroxide flows. Water is added to the catholyte cycle, which is used up in the production process to produce hydroxyl ions and hydrogen gas. The hydroxyl anions and sodium cations formed in this way. Typically, this solution is recirculated before it is discharged from the electrolyser with the aim of a concentrate of between 32-35%. A 50% concentration is possible by evaporation of a sodium hydroxide solution using steam.
The advantage of membrane electrolysis over mercury technology
Obtaining sodium hydroxide using membrane electrolysis has many benefits compared to the mercury method. These are, among others:
- operation at a lower clamping voltage compared to mercury cells,
- lower consumption of electrical energy and energy in the form of steam,
- the ability to change the current load on a daily basis, which allows you to take full advantage of the cheaper night tariff,
- minimising the risk of environmental contamination.
Is the mercury method safe?
In this method, mercury is the cathode material. It takes part in the electrolysis process in which caustic soda lye is formed. Unfortunately its use is very dangerous because mercury:
- acts destructively on the human body – it accumulates in the kidneys and damages them, destroys biological membranes and connects with the proteins of the body, disrupting biochemical processes; mercury poisoning can lead to death, due to respiratory or circulatory failure or mucositis,
- causes environmental contamination – during the production of sodium hydroxide, there are mercury losses that enter the water, air, soil, waste and the product itself.
It is for the above-mentioned reasons that the use of the mercury method in the production of soda lye has become less and less popular for membrane electrolysis around the world. The membrane technology is a method of obtaining high quality sodium hydroxide with regard to the ecological and economic aspects.
